
When the reaction begins, the concentration of A and B starts reducing while the concentration of C and D increases. 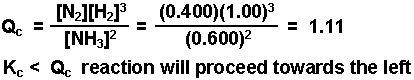
Here A and B are reactants while C and D are products. Notice that the equilibrium concentration of hydrogen bromide is still higher than the equilibrium concentrations of hydrogen gas and bromine gas, as you would expect for an equilibrium for which #K_(eq) > 1#.Let’s consider a reversible equation for understanding equilibrium in chemical reactions The values are rounded to three sig figs. This means that the equilibrium concentrations of the three chemical species that take part in the reaction will be Notice that in order to avoid having negative equilibrium concentrations for hydrogen gas and bromine gas, you must pick the positive value of #x#. Now, this quadratic equation will produce two solutions, one positive and one negative. #color(purple)("E")color(white)(aacolor(black)(0.300+x)aaaacolor(black)(0.150+x)aaacolor(black)(1.20 - color(red)(2)x)#īy definition, the equilibrium constant will be equal to Set up an ICE table using the initial concentrations 
Similarly, the equilibrium concentration of hydrogen bromide will be lower than its initial value. 
As a result, the equilibrium concentrations of hydrogen gas and of bromine gas will be higher than their respective initial values. Now, because #Q_c > K_(eq)#, the equilibrium will shift to the left, i.e. #Q_c = (_t)/(_t * _t) -> # does not use equilibrium concentrations! you should expect the equilibrium concentration of hydrogen bromide to be higher than the equilibrium concentrations of hydrogen gas and bromine gas.īefore moving on to the calculations, use the given values to calculate the reaction quotient, #Q_c#, which can be used to determine the direction in which the equilibrium will proceed when these concentrations of reactants and product are mixed together. Notice that you have #K_(eq) > 1#, which means that at this given temperature, the forward reaction will be favored, i.e. Moreover, you also know the value of the equilibrium constant, #K_(eq)#, which is said to be equal to #11.2#. The problem provides you with the initial concentrations of hydrogen gas, #"H"_2#, bromine gas, #"Br"_2#, and hydrogen bromide, #"HBr"#. the same rule still applies in terms of ICE tables?īut yes, an ICE table is what you need to use here in order to find the answer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
